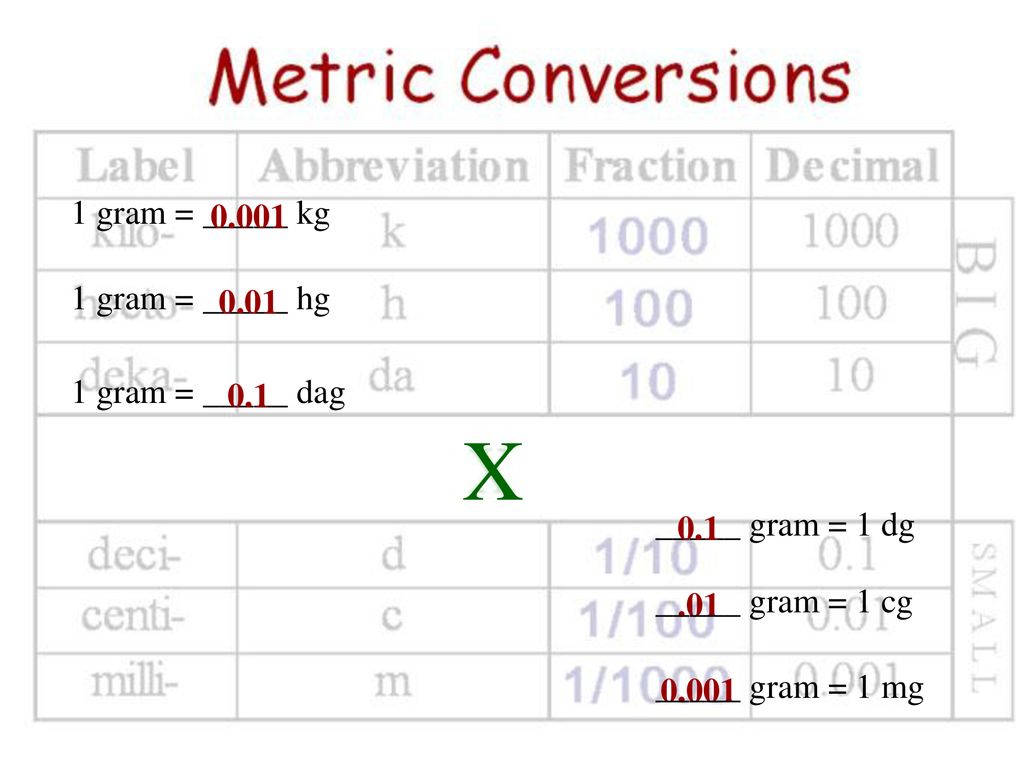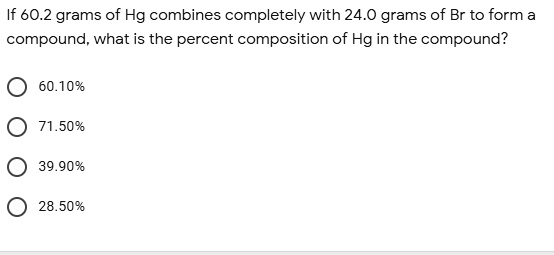
SOLVED: If 60.2 grams of Hg combines completely with 24.0 grams of Br to form compound, what is the percent composition of Hg in the compound? 60.10% 71.50% 39.90% 28.50%

5 gram each of the following gases at 87^∘C and 750 mm pressure is taken. Which of them will have the least volume?

SOLVED:What is the mass in grams of a single atom of each of the following elements: (a) Hg, (b) Ne?

![50% OFF [DECEMBER 30 2022 EXPIRY] HG Flaxseeds (100 grams) – healthmart.ph 50% OFF [DECEMBER 30 2022 EXPIRY] HG Flaxseeds (100 grams) – healthmart.ph](http://cdn.shopify.com/s/files/1/0380/8244/5445/products/falx-min.jpg?v=1626241976)